Draw Sodium Atom, Web this video shows how to draw the orbital diagram of sodium (na).
Draw Sodium Atom - Web in this video we'll look at the atomic structure, valence electrons, ionic charge, and ionic bonding for the sodium atom. Sodium has 2 electrons in its first shell, 8 in its second and 1. Web draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom. Web sodium, the second alkali metal in the first group of periodic table is highly chemically reactive. So, an atom's outer electrons are usually the most important in chemistry. Web the sodium atom loses its outer electron to become a sodium ion. When we write the configuration we'll put all 11. Web in order to write the na electron configuration we first need to know the number of electrons for the na atom (there are 11 electrons). Niels bohr proposed an early model of the atom as a central nucleus containing protons and. It also shows how to write the electron configuration of sodium (na) and the shorthand nobl. 11), the most common isotope of the element sodium. Web sodium is used as a heat exchanger in some nuclear reactors, and as a reagent in the chemicals industry. So the group number corresponds to how. Web sodium, the second alkali metal in the first group of periodic table is highly chemically reactive. Web electrons in the outermost shell of. Web the sodium atom loses its outer electron to become a sodium ion. Web the electron configuration of sodium refers to the arrangement of electrons in the sodium atom’s orbitals. (generally, the least electronegative element should be placed in. The sodium ion still has 11 protons ( 11 positive charges) but now only 10 electrons ( 10 negative charges). Web. It also shows how to write the electron configuration of sodium (na) and the shorthand nobl. I show you where sodium is on the periodic table and how to determine how many valence electrons it has. Niels bohr proposed an early model of the atom as a central nucleus containing protons and. (generally, the least electronegative element should be placed. It describes how electrons are distributed among the various atomic. But sodium salts have more uses than the metal itself. Web sodium, the second alkali metal in the first group of periodic table is highly chemically reactive. Web in this video we'll look at the atomic structure, valence electrons, ionic charge, and ionic bonding for the sodium atom. Web the. (generally, the least electronegative element should be placed in. Web one sodium (na) atom gives it's electron to one chlorine (cl) atom. Web in this video we'll look at the atomic structure, valence electrons, ionic charge, and ionic bonding for the sodium atom. 11), the most common isotope of the element sodium. But sodium salts have more uses than the. Web sodium, the second alkali metal in the first group of periodic table is highly chemically reactive. Web electrons in the outermost shell of an atom are most easily transferred or shared with other atoms. Web one sodium (na) atom gives it's electron to one chlorine (cl) atom. When we write the configuration we'll put all 11. Sodium is happy. Web in order to write the na electron configuration we first need to know the number of electrons for the na atom (there are 11 electrons). It also shows how to write the electron configuration of sodium (na) and the shorthand nobl. Web sodium is used as a heat exchanger in some nuclear reactors, and as a reagent in the. Web sodium, the second alkali metal in the first group of periodic table is highly chemically reactive. But sodium salts have more uses than the metal itself. Web sodium is used as a heat exchanger in some nuclear reactors, and as a reagent in the chemicals industry. It also shows how to write the electron configuration of sodium (na) and. Web in order to write the na electron configuration we first need to know the number of electrons for the na atom (there are 11 electrons). I show you where sodium is on the periodic table and how to determine how many valence electrons it has. It describes how electrons are distributed among the various atomic. It also shows how. It also shows how to write the electron configuration of sodium (na) and the shorthand nobl. 61k views 4 years ago. Web the electron configuration of sodium refers to the arrangement of electrons in the sodium atom’s orbitals. Chlorine then has the eight electrons in its outer shell to make it happy. Web for example, if we look at elements. Web electrons in the outermost shell of an atom are most easily transferred or shared with other atoms. Sodium has 2 electrons in its first shell, 8 in its second and 1. 61k views 4 years ago. Web sodium, the second alkali metal in the first group of periodic table is highly chemically reactive. Web one sodium (na) atom gives it's electron to one chlorine (cl) atom. I show you where sodium is on the periodic table and how to determine how many valence electrons it has. Web the electron configuration of sodium refers to the arrangement of electrons in the sodium atom’s orbitals. Web sodium is used as a heat exchanger in some nuclear reactors, and as a reagent in the chemicals industry. Web in order to write the na electron configuration we first need to know the number of electrons for the na atom (there are 11 electrons). The sodium atom (na) is commonly used for examples and. Chlorine then has the eight electrons in its outer shell to make it happy. Web draw the bohr diagram of an atom with 18 electrons or fewer. Web for example, if we look at elements in the first group, like hydrogen, lithium, or sodium, the first group all have one valence electron. When we write the configuration we'll put all 11. Sodium is widely used in pharmaceutical industries to manufacture lifesaving. The sodium ion still has 11 protons ( 11 positive charges) but now only 10 electrons ( 10 negative charges).:max_bytes(150000):strip_icc()/sodiumatom-58b602715f9b5860464c7a22.jpg)
Atom Diagrams Electron Configurations of the Elements
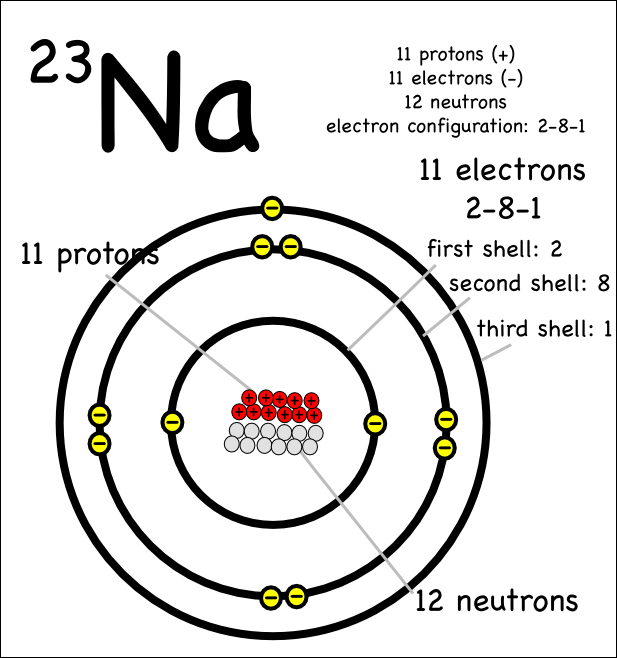
Drawing Atoms Montessori Muddle
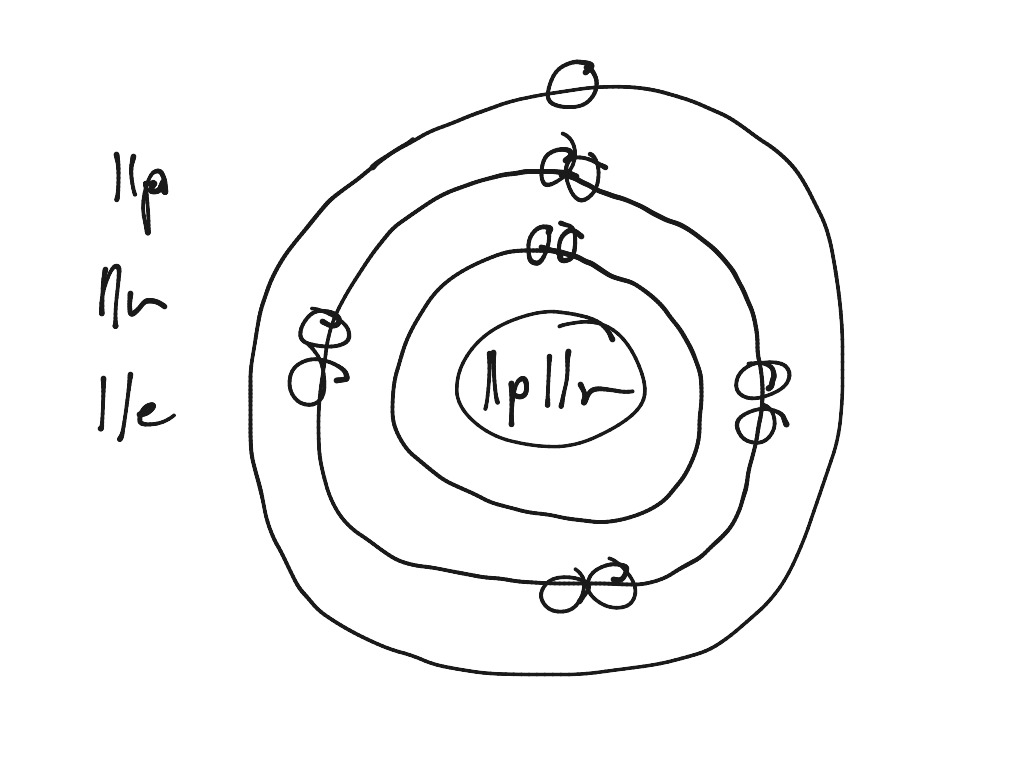
Sodium atom drawing Science, Chemistry, Elements, Atoms, Model
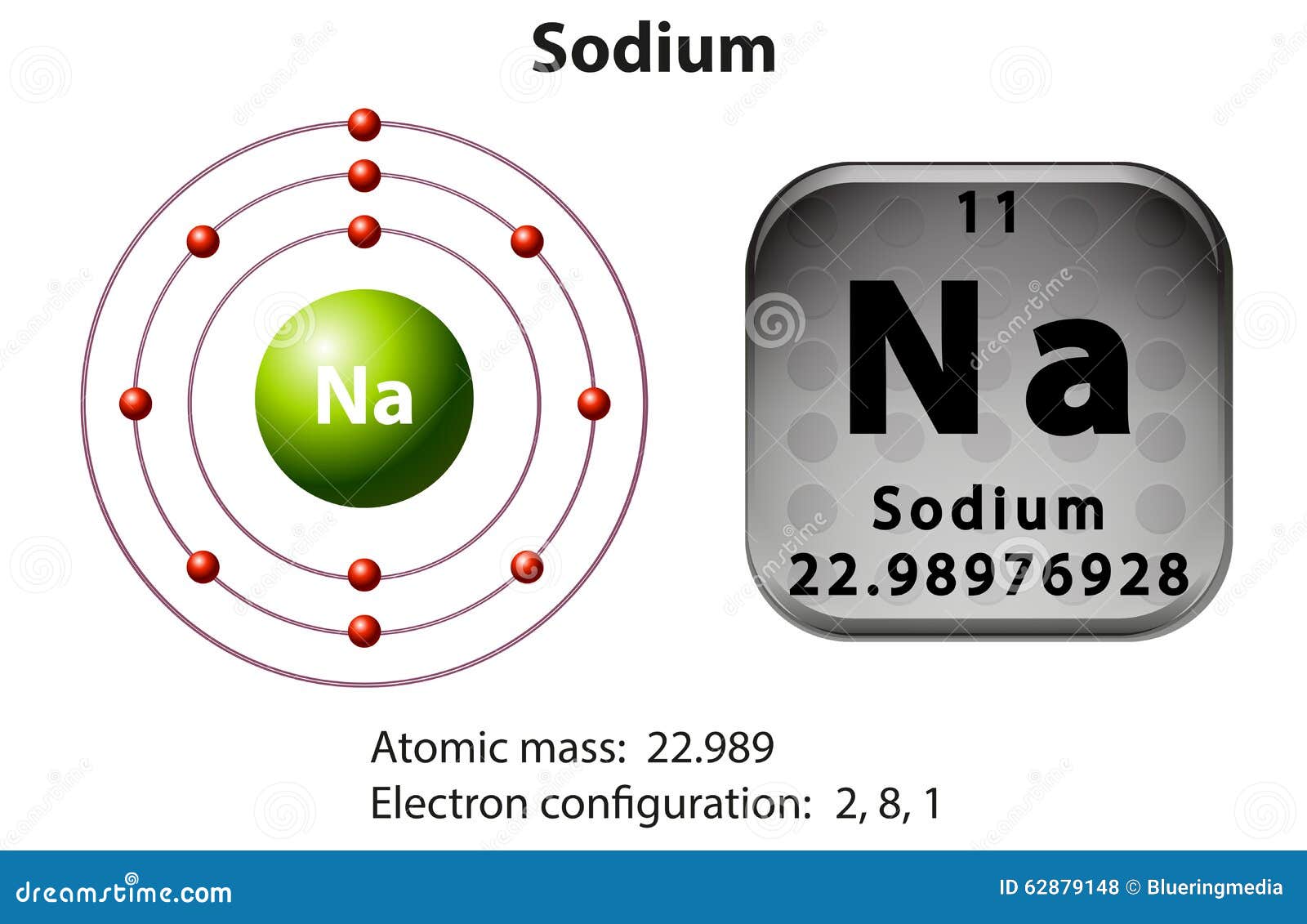
Symbol And Electron Diagram For Sodium Stock Vector Image 62879148

Perfect Tips About How To Draw Sodium Atom Effectsteak33

Atom Sodium Model Vector & Photo (Free Trial) Bigstock
Chemistry 2.Draw the atomic structure of a sodium atom and a sodium
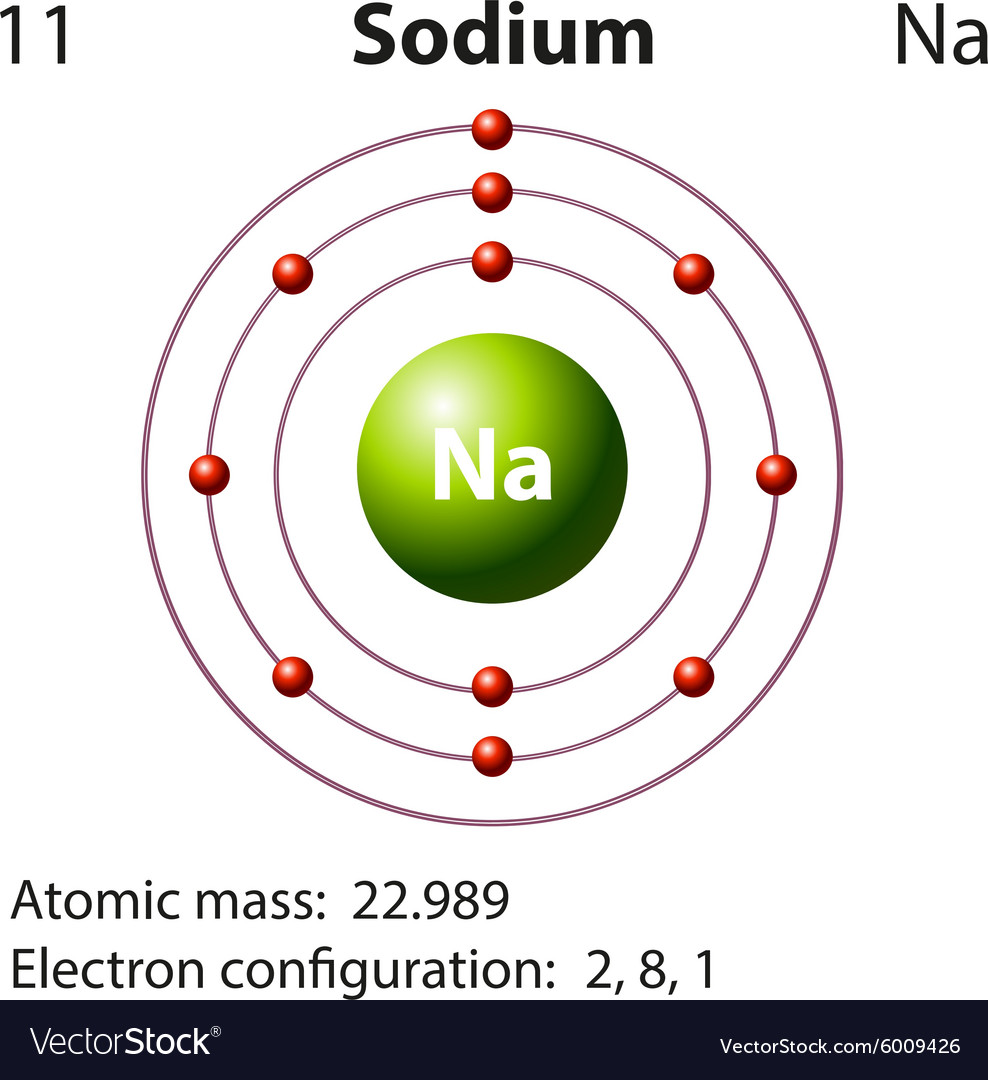
Sodium Atomic Structure

FileElectron shell 011 sodium.png Wikimedia Commons
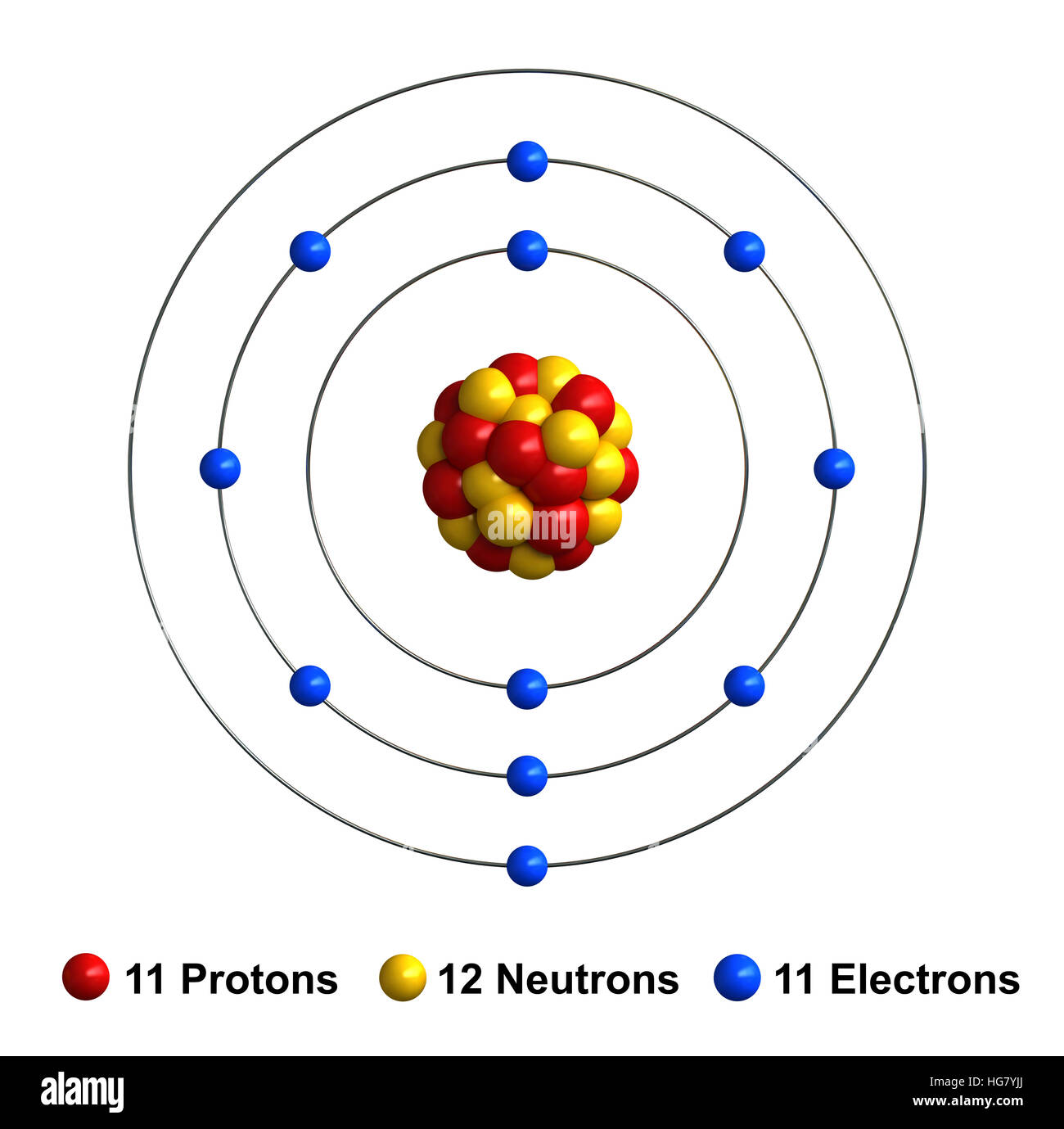
3d render of atom structure of sodium isolated over white background
Web This Video Shows How To Draw The Orbital Diagram Of Sodium (Na).
11), The Most Common Isotope Of The Element Sodium.
So The Group Number Corresponds To How.
It Also Shows How To Write The Electron Configuration Of Sodium (Na) And The Shorthand Nobl.
Related Post: